Everything in this world is made up of atoms. YES, even the writer who created this facts about Atoms list is made up of them.
For thousands of years, humans were puzzled by the existence of these particles and have been studying it for many years.
Even just about 2 centuries ago, we didn’t have any clear understanding of Atoms and anything close to it.
However, now we know a lot more about it and as a result, we will be exploring some incredible atom facts in this post.
Cool Atom facts list
1. An atom is the smallest unit of matter
Simply put, an atom is the smallest unit of any ordinary matter that contains the properties of the very matter.
Almost every matter is made up of atoms. Atoms are the main building block of a matter. Now, let’s make it simpler for you.
To do that, we will assume a situation here that is not practically possible for us. But theoretically, we can assume it.
Let’s say you are dividing a piece of matter as many times as possible. In this instance, you will reach a point where if you could divide the matter anymore, it would lose the properties of the matter itself.
And, this is when you have found the atom of that particular matter. Eventually, any matter is made up of trillions and trillions of individual atoms.
2. Origin of the Idea of Atom

The word Atom actually comes from the Greek word ‘Atomos’. ‘Atomos’ means ‘Indivibisible’/‘Uncuttable’.
About 2500 years ago, a philosopher named ‘Leucippus of Miletus’ of Greece has introduced the idea of Atom.
However, his student ‘Democritus’ is regarded as the discoverer of ‘Atomism’. Atomism is the idea that atoms are the smallest unit of any particle.
He also said that Atoms are infinite in number, not divisible, eternal, and uncreated.
3. Types of Atoms

As Atoms are the smallest units of any chemical element, the number of different kinds of atoms depends on the number of chemical elements as well.
As of now, there are 118 chemical elements that are confirmed. That means, there are also 118 kinds of atoms -1 for each chemical element – are known as of right now.
4. What are Atoms Made of
Earlier, most of the scientists and philosophers would think that atoms are indivisible. However, with the advancements in the scientific field, now we know that even atoms are divisible.
If you divide an atom, you will find 3 types of particles. And these are known as the ‘Subatomic Particles’.
These 3 main subatomic particles are called:
- Electron
- Proton
- Neutron
That means atoms are primarily made of 3 subatomic particles those are Electron, Proton, and Neutron.
Along with that according to scientists, there are over 200 different types of subatomic particles.
5. What are Subatomic Particles Made of
Interestingly, after the discovery of the subatomic particles, scientists believed that it is impossible to divide the subatomic particles.
As a result, these were known as the ‘Fundamental Particles’. However, after a while, it was discovered that even the subatomic particles i.e., Proton and Neutron are also divisible.
To be noted, you can’t divide an electron. We will talk about that in another section down below.
Now, when you divide some of the subatomic particles, you will find ‘Quarks’. That means the ‘Quarks’ are actually the elementary/fundamental particles.
6. How Atoms were Created

As almost every matter in the universe is made up of individual atoms, they had to be formed during the Big Bang.
Now, theoretically, atoms were made about 13.7 billion years ago after the Big Bang. During the Big Bang, the universe was so hot that it was not suitable for forming atoms.
However, over time, the universe cooled down. And the condition of the universe was ideal for forming quarks (protons and neutrons) and electrons.
Then, with the combination of electrons, protons, and neutrons; atoms began to form.
7. Can You Observe/See Atom?

To put it simply, you cannot see or observe atoms with the naked eye. These are incredibly tiny particles. Interestingly, atoms are so small that it is not even possible to see those using typical optical microscopes.
As a result, scientists from around the world have been trying to develop a microscope that can be used to see individual atoms.
8. IBM and Their Scanning Tunneling Microscope

Speaking of inventing microscopes for observing atoms, researches at IBM have invented a unique type of microscope named the ‘Scanning Tunneling Microscope’.
With the help of this microscope, it was possible to observe atoms for the first time.
The ‘Scanning Tunneling Microscope’ was invented in 1981. The inventor of this device was awarded the Nobel Prize.
Nowadays, there are lots of ‘Nanoscale Microscopes’ or ‘Electron Microscopes’ with which it is possible to see atoms.
9. Structure of Atom
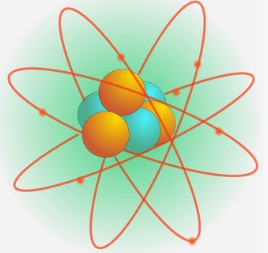
As we have previously said, an atom consists of 3 basic subatomic particles named electron, proton, and neutron.
Here, the proton and the neutron resides at the core of the atom. The core is called the nucleus of an atom.
Now, where do the electrons go? Interestingly, the electros will not be at the nucleus. Rather, the electrons will be at the electron shells surrounding the nucleus.
10. Atoms Are Mostly Empty Space

You might have heard this a lot before that atom is almost empty space. But how much is it? Matter of fact, it is almost all of it.
As you know that the core of the atom contains both the proton and the neutron. And, the electrons are in the outer region surrounding the nucleus.
Now, there is a huge gap between the nucleus or the core and the electrons at the outer region. And, that is the empty space in an atom.
For instance, the empty space on a Hydrogen atom is about 99.9999999999996%. That means the rest of the 0.0000000000004% of the Hydrogen atom is full.
11. Confusion Between Atoms and Molecules
Although a lot of us get confused between ‘Atoms’ and ‘Molecules’, but these are 2 totally unique things.
Simply put, when a group of atoms combines or bond together, then it is called a molecule. That means molecules are created when multiple atoms are combined.
12. Size of Atoms

You know that atoms are really small. But how small is it? They are so small that, you will need trillions of atoms to make anything.
Also, the size of the atom actually differs from element to element. However, the size of a typical atom would be about 0.3 Nano feet across.
This is so small that you will have a hard time even imagining it. So, here’s an analogy for you.
If you are 6 ft. tall and the size of an atom is about 0.3 Nano feet, then the atom is about 20 billion times smaller compared to you. Let that sink in for a moment.
More to that, the approximate diameter of the Earth is about 12000 kilometers. Now, compared to that if you are 6 feet tall, then Earth is about 6 million times bigger than you. So, it is evident that atoms are unimaginably small.
13. The Largest, The Smallest, The Lightest, and The Heaviest Atom

By mass, Hydrogen atom is the lightest. And, Uranium is the heaviest atom by mass among the naturally occurring elements.
However, by size, the smallest atom is the Helium atom. And, Francium is one of the atoms that have the largest atomic radius.
14. 9 Atom and The Human Body

Yes, we are also made of atoms. Do you know how many atoms are in your body? Believe it or not, there are about ‘7 billion billion billion’ atoms in the body of an adult human of 70 kg.
That is 27 zeros after a 7! Yes, you have heard it right. On top of this, our body replaces about 98% of all the atoms every year.
15. Electrons, Protons, and Neutrons
You know that atoms are made of subatomic particles. There are 2 types of subatomic particles such as elementary and composite particles.
The elementary particles are indivisible. And the composite particles are made of other smaller particles known as ‘Quark’.
Now, the electron is an elementary particle. It is a negatively charged particle. On the other hand, protons are positively charged subatomic particles.
And, neutrons are electrically neutral particles. They don’t have any electric charges.
16. Number of Atoms in the Known Universe

You might have already guessed that there would be a great load of atoms in the entire universe.
But how many Atoms are there in the universe? There is no precise answer to this question.
However, researchers suggest that there are about 10^78 to 10^82 atoms in the observable universe. Unfathomable, isn’t it?
17. Splitting an Atom
It was ‘John Cockcroft’ and ‘Ernest Walton’ who split the atom for the first time in history.
It was back in 1932. They split the lithium nucleus into 2 helium nuclei. Later, in 1938, other 2 scientists from Germany named ‘Otto Hahn’ and ‘Fritz Strassmann’ split atoms of Uranium into multiple parts.
It was the first time when we have split an atom of a heavy element.
18. Neutron-less Element
We know that atom is made up of 3 basic subatomic particles named electron, proton, and neutron.
However, in the case of Hydrogen, it is not applicable. Hydrogen is the only atom that doesn’t contain any neutron. Rather, it contains a positively charged proton and a negatively charged electron.
19. Atom and Chemical Elements
You already know the definition of an atom. But what are elements? We tend to mix up between these two.
For your information, elements are the substances that contain only one kind of atom. As previously mentioned, there are about 118 confirmed elements.
Interestingly, not all of these elements are naturally occurring. Among these, there are about 24 elements that are manmade.
That means you will not find these elements in nature. Rather, we have created them in the laboratories under certain conditions.
20. Most Abundant Chemical Elements in the Universe

It was after 380,000 years of the Big Bang, the universe was cooled down to be an ideal place for forming atoms. And, only then, the atoms could start to form new chemical elements.
Reports show that the first atoms were Hydrogen and Helium. Still, Hydrogen is the most abundant element in the universe.
The second most abundant element is Helium. And the third most abundant element in the universe is Oxygen.
21. Most Abundant Element on Earth

The Universe might have Hydrogen and Helium in the most abundant amount. However, the Earth is a bit different here.
The most abundant element on Earth is actually Oxygen. It constitutes about 46.6% of Earth’s mass. Next up, Silicon is the 2nd most abundant element on Earth.
22. The Father of Atomic Theory

The idea or theory that every object in the world/universe is made up of many individual tiny particles was prevalent from the ancient time.
However, it was John Dalton who included the idea of atoms in the modern science field for the first time.
He was an English chemist, physicist, meteorologist, and more. His atomic theory was the first in history which attempted to describe the structure of matters in terms of atoms. As a result, he is regarded as the father of modern atomic theory.
23. How Many Atoms are there On Earth

You know that atoms are incredibly small. For your information, you can fit in around 1 million of carbon atoms across a typical human hair.
Now, from the mass of Earth and the average mass of an atom, we can find out that the total number of atoms on Earth.
From that calculation, we know that there are about 10^50 atoms in our planet.
24. Atoms vs. Cell

What is an atom? Or what is a cell? We commonly confused between these two terms as well.
| Atom | Cell |
| Make up molecules | Made of molecules |
Simply put, cells are made of billions and billions of molecules. And, you know that molecules are made up of atoms.
However, cells are living things but atoms are non-living things. That means atoms are very smaller than cells.
Matter of fact, reportedly, there are about 100 trillion atoms in an average size cell.
25. We are Stardust

By mass, about 99 percent of the human body is made up of 6 elements such as:
- Oxygen
- Carbon
- Hydrogen
- Nitrogen
- Calcium
- Phosphorus
And, the other percentage is made up of sulfur, potassium, chlorine, magnesium, etc. Now, you know that all the elements – including those we are made of – are actually made up of atoms.
And these atoms were created after the Big Bang. That means we are actually made of the same elements that were created after the Big Bang and are still in the space and stars.
As most of the elements that make up our bodies were formed in the stars, we can say that we are actually stardust.
Last Words
Atom is the building block of matter. At the moment, we know a lot about atoms. In the olden times, humans could only think about the idea of atoms.
However, with the innovations in science and technology, now, we can even see atoms with our own eyes.
We hope this informational post will come in handy for you. If you have liked the post, you can share it with others.
Have you enjoyed this post about Atoms? Then we would recommend you read this post on facts about Black Holes. And, don’t forget to leave your thoughts and opinions in the comments below!
